TPHA ( 72503 )
TPHA Kit (72503 / 72504) is a passive haemagglutination assay designed for qualitative and semi-quantitative detection of antibodies to Treponema pallidum in human serum or plasma. This kit is ideal for syphilis screening, blood donor testing, and clinical diagnostic support.
Key Features
- Detection Method: Passive Haemagglutination (TPHA)
- Detects antibodies to Treponema pallidum
- Sample Type: Human Serum or Plasma
- Qualitative & Semi-Quantitative Testing
- Available Formats: 200 Tests (72503) / 500 Tests (72504)
- High Specificity: 99.72%
- High Sensitivity: 100%
- Storage: 2–8°C
- Includes Positive & Negative Controls
- Suitable for Blood Donor Screening
Description
TPHA Kit (72503 / 72504)
The TPHA Kit (72503 / 72504) is designed for qualitative and semi-quantitative detection of antibodies to Treponema pallidum in human serum or plasma using passive haemagglutination technology. This test is widely used for syphilis screening, blood donor testing, and clinical diagnostic applications.
The TPHA assay provides reliable detection of treponemal antibodies, making it an essential tool for laboratory diagnosis of syphilis infection.
Product Overview
- Product Name: TPHA Kit
- Product Codes: 72503 (200 tests), 72504 (500 tests)
- Method: Passive Haemagglutination
- Sample Type: Human Serum / Plasma
- Application: Syphilis antibody detection
- Storage Temperature: 2–8°C
- Intended Use: In vitro diagnostic use
Intended Use
The TPHA kit is intended for:
- Qualitative detection of Treponema pallidum antibodies
- Semi-quantitative antibody titration
- Blood donor screening
- Diagnostic support for suspected syphilis infection
The test detects antibodies present in human serum or plasma samples.
Test Principle
The TPHA test uses preserved avian erythrocytes coated with Treponema pallidum antigens:
- Positive samples show agglutination pattern
- Negative samples show cell settling (button formation)
Agglutination patterns can be interpreted visually or using a plate reader.
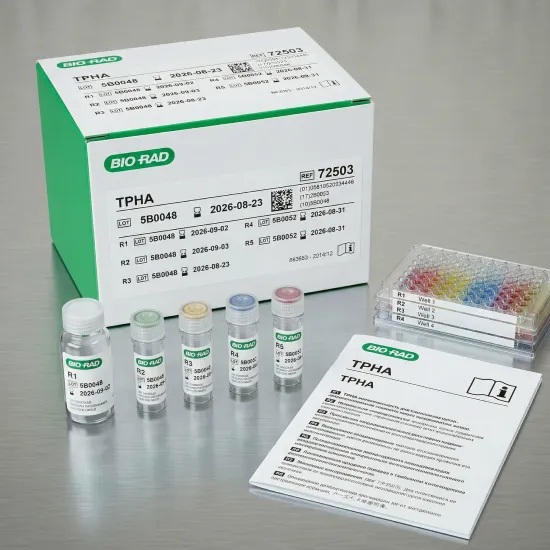
Kit Components
The TPHA kit includes:
- Test Cells (R1) – Antigen coated erythrocytes
- Control Cells (R2)
- Diluent (R3)
- Positive Control (R4)
- Negative Control (R5)
These components ensure accurate and validated test results.
Sample Requirements
Compatible sample types include:
- Human serum
- Plasma (EDTA, Sodium Citrate, Heparin, ACD)
Sample storage:
- 2–8°C for up to 7 days
- -20°C for long-term storage
Assay Procedure Overview
Qualitative Assay
- Dilute sample
- Add test cells
- Incubate at room temperature
- Read agglutination pattern
Semi-Quantitative Assay
- Serial dilution method
- Determine antibody titer
- Detection range: 1:80 to 1:10240
Performance Characteristics
Sensitivity
- 100% sensitivity (435/435 samples)
Specificity
- 99.72% specificity (5031 samples tested)
Analytical Sensitivity
- 0.05 IU/mL detection sensitivity
Applications
- Syphilis screening
- Blood bank screening
- Clinical diagnosis
- Laboratory testing
- Epidemiological studies
- Hospital laboratories
Storage and Stability
- Store at 2–8°C
- Do not freeze
- Stable until expiry date when stored properly
Advantages
- High sensitivity and specificity
- Reliable haemagglutination method
- Qualitative and semi-quantitative results
- Easy interpretation
- Suitable for high-throughput screening
- Includes controls for validation
Technical Specifications
| Parameter | Details |
| Product Name | TPHA Kit |
| Method | Passive Haemagglutination |
| Sample Type | Serum / Plasma |
| Test Format | 200 / 500 Tests |
| Storage | 2–8°C |
| Sensitivity | 100% |
| Specificity | 99.72% |
Diagnostic Notes
- Positive results indicate presence of Treponema pallidum antibodies
- Negative results do not exclude early infection
- Confirmatory testing recommended when required
The TPHA Kit (72503 / 72504) provides a reliable, accurate, and efficient solution for syphilis screening and diagnosis, making it an essential diagnostic tool for clinical laboratories and healthcare institutions.